Customized Integrated Solutions
Integrated Hydrogen Peroxide Biodecontamination Systems for Cleanrooms, Laboratories, and Controlled Environments
Flexible biodecontamination integration—from portable system coordination to fully automated facility infrastructure.
Many controlled environments require more than portable biodecontamination alone. Pharmaceutical cleanrooms, biosafety laboratories, vivariums, and research facilities often need repeatable, validated decontamination cycles that integrate with facility operations.
CURIS integrated biodecontamination systems use Hybrid Hydrogen Peroxide™ technology to deliver vaporized hydrogen peroxide either directly within a space or through fixed injection points—while enabling increasing levels of automation, control, and facility coordination.
The CURIS® Integration Architecture™ defines a clear framework for implementing biodecontamination based on your facility’s operational needs—from simple system coordination to fully automated, facility-wide biodecontamination.
What is an Integrated Biodecontamination System?
An integrated biodecontamination system uses vaporized hydrogen peroxide to deliver controlled, repeatable decontamination cycles within a room, chamber, or controlled environment.
Depending on the system configuration, vapor may be:
- Delivered directly from a portable generator within the space
- Introduced through fixed injection points from outside the room
- Coordinated with facility systems such as HVAC, door interlocks, and automation controls
Integrated systems are designed to reduce variability, improve repeatability, and align decontamination with facility workflows.
Where Integrated Biodecontamination Systems Are Used
Why Facilities Move to Integrated Biodecontamination
Facilities adopt integrated biodecontamination to improve:
- Material throughput and cycle efficiency
- Turnaround time between operations
- Labor requirements and operator dependency
- Consistency and validation of contamination control
In high-throughput environments, automated biodecontamination systems can significantly reduce bottlenecks associated with manual cleaning processes.
Integrated hydrogen peroxide vapor biodecontamination systems are widely used across regulated and research environments:
- Pharmaceutical cleanrooms: aseptic processing, fill-finish manufacturing, isolators
- Biosafety laboratories (BSL-2 to BSL-4): room and equipment decontamination
- Vivarium facilities: animal rooms, cage wash, and transfer spaces
- Biotechnology laboratories: cell and gene therapy research environments
- Containment research facilities: high-risk and specialized applications
- Material airlocks (MALs) and chamber-based decontamination systems
These environments require consistent, documented contamination control processes to protect product quality, research integrity, and personnel safety.
Why Facilities Move to Integrated Biodecontamination
Compared to manual disinfection or portable-only approaches, integrated systems enable:
- More repeatable and validated cycles
- Reduced operator variability
- Improved process consistency
- Better documentation and reporting
- Alignment with facility automation and safety systems
How Integrated Biodecontamination Works
Facility Considerations for Integrated Systems
Implementing an integrated biodecontamination system may require coordination with facility infrastructure, including:
- HVAC operation and airflow control
- Room pressurization and isolation during cycles
- Door sealing and interlock systems
- Environmental conditions such as humidity
Depending on the level of integration, CURIS can:
- Provide system design requirements
- Collaborate with facility and engineering teams
- Support full turnkey integration when needed
CURIS® Integration Architecture™
Three Levels of Integrated Biodecontamination
Integrated biodecontamination is not a one-size-fits-all approach. Facilities vary in their need for automation, infrastructure, and operational control.
The CURIS platform supports this range through three defined levels of integration—allowing facilities to implement what they need today while maintaining a clear path to more advanced automation.
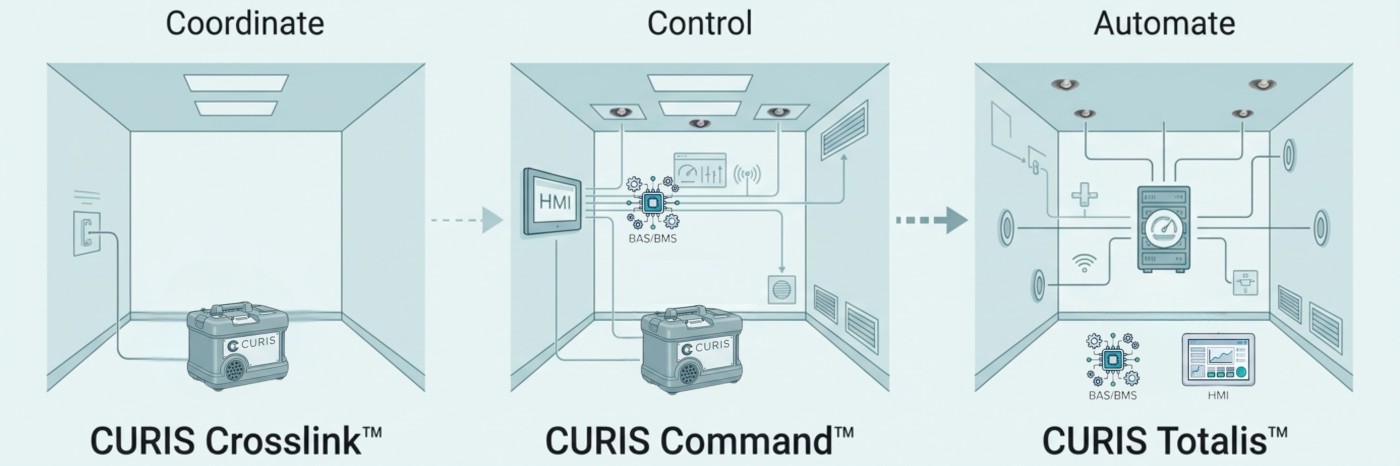
CURIS Crosslink™—Coordinated Integration
Entry point to integrated biodecontamination
CURIS Crosslink™ represents the first level of integration—bringing biodecontamination into alignment with facility workflows while maintaining the flexibility of a portable-powered system.
Best for:
Facilities that need coordinated biodecontamination with existing workflows—without adding permanent infrastructure. Common applications include chamber-based material transfer systems.
Key Features:
- Portable-powered system deployable inside or outside the room
- Enables coordination with facility workflows and operational procedures
- Optional communication with facility systems (e.g., HVAC response, door interlocks)
- Supports repeatable cycle execution without fixed infrastructure
CURIS Command™—Controlled Integration
Structured control and automation of biodecontamination
CURIS Command™ builds on coordinated systems by introducing centralized control, automation, and repeatable process execution.
Best for:
Facilities that require structured control, automation, and repeatable cycle execution across rooms or processes.
Key Features:
- Centralized control via integrated HMI and system logic
- Pre-programmed, repeatable biodecontamination cycles
- Coordinated control of facility systems (HVAC, doors, safety interlocks)
- Enhanced cycle documentation and reporting
CURIS Totalis™—Fully Automated, Lights-Out Biodecontamination
Fixed, whole-space or facility-wide biodecontamination
CURIS Totalis™ represents full system integration—embedding biodecontamination directly into facility infrastructure.
Best for:
Facilities seeking fully automated, facility-wide biodecontamination integrated directly into building infrastructure.
Key Features:
- Permanently installed biodecontamination infrastructure
- Full integration with BAS/BMS systems for automated operation
- Centralized control across rooms, suites, or entire facilities
- Supports lights-out or operator-initiated automation
- Enables high-throughput material transfer workflows
- Reduces manual intervention across facility operations
Integration With Facility Infrastructure
Depending on the integration level, CURIS systems can interact with:
- Building automation systems (BAS/BMS)
- HVAC and air handling systems
- Door interlocks and access controls
- Environmental monitoring systems
- Reporting and compliance platforms
This allows facilities to align biodecontamination with operational workflows and safety requirements.
Designed for Controlled Environments
CURIS systems support a wide range of controlled environments:
Pharmaceutical & Biopharmaceutical
- Cleanrooms and aseptic suites
- Fill-finish and manufacturing
- Isolators and material airlocks
Biosafety & Containment Labs
- BSL-2, BSL-3, BSL-4 laboratories
- Infectious disease research environments
Vivarium Facilities
- Animal holding rooms
- Cage wash and transfer areas
Biotechnology & Research
- Cell and gene therapy labs
- Translational research facilities
Across these environments, CURIS provides a consistent biodecontamination technology platform aligned with facility operations.
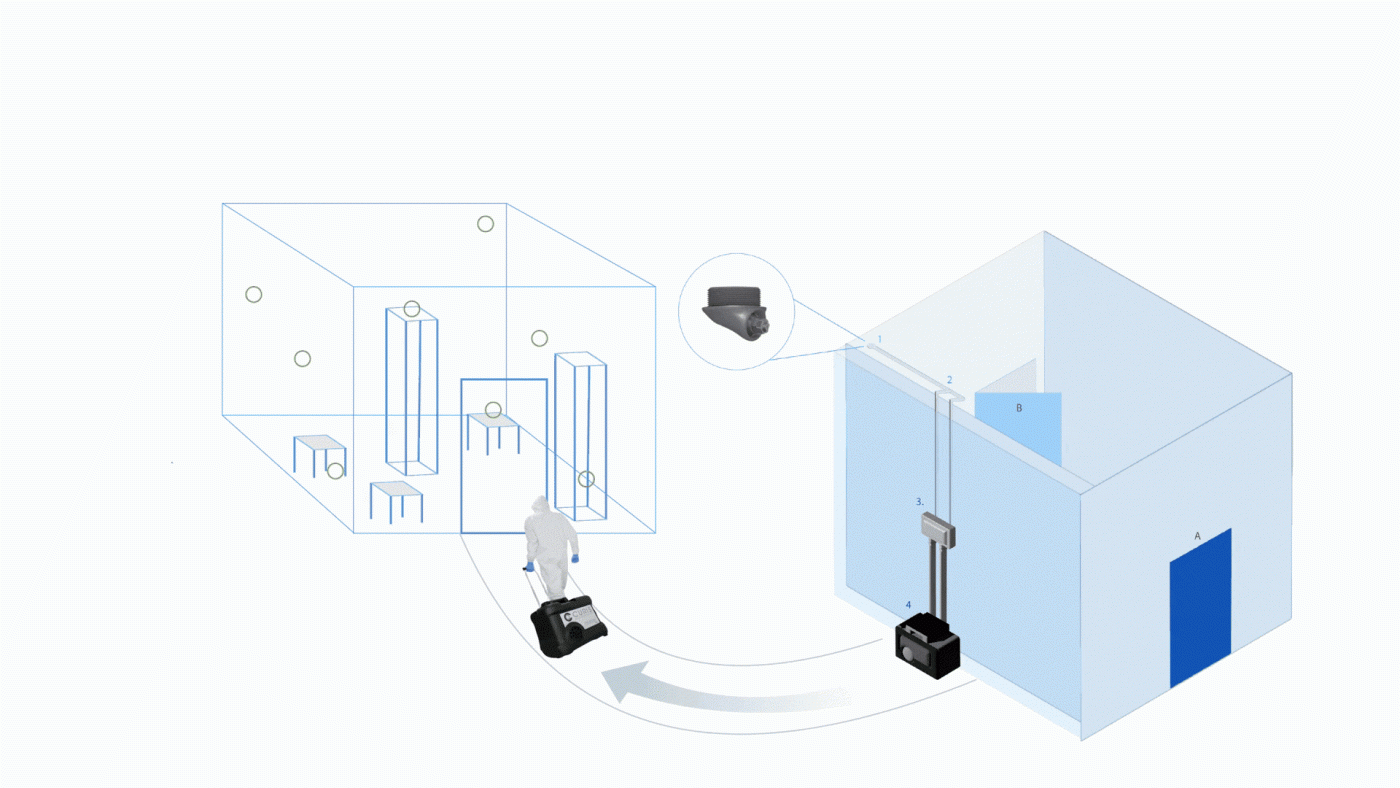
Room Decontamination for Biosafety Laboratories and Vivariums
Biosafety and vivarium environments often require room-level biodecontamination to protect personnel, research integrity, and animal health.
Integrated systems support:
- Whole-room decontamination cycles
- Equipment and chamber decontamination
- Coordinated workflows with safety systems
By delivering vapor through controlled methods and supporting repeatable cycles, CURIS systems help maintain consistent contamination control procedures.
Flexible Integration for Real-World Facilities
Designed for Real-World Facility Constraints
Every facility presents unique challenges—from material compatibility to workflow requirements.
Some environments may require:
- Multiple transfer strategies
- Separate workflows for sensitive materials
- Coordination with existing facility classifications
The CURIS® Integration Architecture™ is designed to accommodate these variations through flexible system configurations.
Every facility operates differently. Some prioritize portability and flexibility. Others require automation and centralized control.
The CURIS platform supports both.
- Crosslink™ enables coordinated operation with minimal infrastructure
- Command™ adds structured control and automation
- Totalis™ delivers full facility integration and automation
This approach allows facilities to implement the level of integration that fits their operational needs—without overengineering or oversimplifying the solution.
Frequently Asked Questions
What is an integrated biodecontamination system?
A system that delivers vaporized hydrogen peroxide in a controlled, repeatable manner, often integrated with facility infrastructure.
What is the difference between portable and integrated systems?
Portable systems operate independently within a room, while integrated systems coordinate with facility systems or use fixed infrastructure, such as wall or ceiling mounted nozzles, for improved consistency and automation.
Can CURIS systems integrate with building automation systems?
Yes. Depending on the system level, CURIS solutions can communicate with or control BAS/BMS systems, HVAC, and facility controls.
Do all systems require fixed installation?
No. CURIS Crosslink™ and Command™ systems are portable-powered or docked in place, while Totalis™ is fully fixed.
Request an Integration Assessment
Understand which level of integration is right for your facility.
Speak with a CURIS specialist to evaluate your environment, workflows, and automation goals.
👉 Talk to a CURIS Integration Specialist