Automated Material Airlocks with CURIS Decontamination Chambers
Integrated Hybrid Hydrogen Peroxide™ Vapor Decontamination for Material Transfer
Improve material throughput, reduce bottlenecks, and achieve repeatable, validated decontamination with automated chamber systems powered by CURIS Hybrid Hydrogen Peroxide™ technology.
Integrated Chamber Biodecontamination for Material Transfer |
Material transfer is one of the most critical—and often limiting—steps in contamination control.
In pharmaceutical cleanrooms, biosafety laboratories, and vivarium environments, manual spray-and-wipe processes introduce variability and increase labor demands.
CURIS decontamination chambers provide automated, repeatable biodecontamination cycles for materials, equipment, and components moving into controlled environments.
As part of the CURIS® Integration Architecture™, chamber systems can be deployed at multiple levels—from controlled chamber operation to fully automated, facility-wide material transfer workflows.
Increase Throughput and Eliminate Material Transfer Bottlenecks |
Manual decontamination processes often become a constraint on facility performance.
CURIS chamber systems help facilities:
- Process higher volumes of materials per cycle
- Reduce reliance on manual wiping and staging processes
- Improve turnaround time between operations
- Minimize delays in production or research workflows
For high-throughput environments, automated chamber-based biodecontamination can significantly improve operational efficiency.
What Is a Biodecontamination Chamber? |
A biodecontamination chamber is an enclosed system that uses vaporized hydrogen peroxide (VHP / VPHP) to disinfect materials before they enter higher-grade or controlled environments.
CURIS chambers use Hybrid Hydrogen Peroxide™ technology, delivering a combination of vapor and micro-aerosolized hydrogen peroxide for consistent surface coverage and sporicidal efficacy.
These systems are commonly used in:
- Material airlocks (MALs)
- Cleanroom material transfer
- Biosafety laboratory workflows (BSL-2 to BSL-4)
- Vivarium material and equipment movement
These systems are increasingly adopted as facilities move toward more automated and repeatable contamination control processes.
Automation Improves Compliance and Contamination Control |
Automated chamber systems support consistent, compliant contamination control by providing:
- Repeatable, validated cycles
- Reduced operator variability and human error
- Improved documentation and compliance readiness
- Safer, controlled handling of incoming materials
Cycle Time and Throughput Performance
Cycle time is a key driver of material throughput, with CURIS chamber systems achieving:
- Validated biodecontamination cycles in under 60–90 minutes
- Faster turnaround with optimized aeration and airflow conditions
This supports more efficient operations and reduced downtime.
Low-Concentration Hydrogen Peroxide for Safer Operation
CURIS systems use a 7% hydrogen peroxide solution, significantly lower than traditional high-concentration systems.
This enables:
- Improved compatibility with sensitive materials and electronics
- Reduced risk to personnel and operators
- Faster aeration and return-to-operation times
- Lower environmental and facility impact
Chamber Integration Within the CURIS® Integration Architecture™
CURIS chamber systems are designed to align with different levels of facility integration.
Controlled Chamber Operation (Command™-Level Integration)
For facilities requiring structured, repeatable processes:
- Pre-programmed, automated biodecontamination cycles
- Integrated HMI touchscreen control
- Cycle monitoring, reporting, and audit tracking
- Coordination with facility systems such as HVAC and door interlocks
Best for:
Facilities seeking controlled, repeatable biodecontamination without full facility automation.
Fully Integrated Chamber Systems (Totalis™-Level Integration)
For facilities requiring advanced automation and throughput:
- Full integration with BAS/BMS systems
- Automated material transfer workflows
- Centralized control across multiple chambers or areas
- Support for lights-out or operator-initiated operation
Best for:
Facilities implementing high-throughput, fully automated contamination control strategies.
Integration with Leading Chamber Manufacturers
In addition to CURIS Integration Architecture™ configurations, systems can also be integrated into third-party chamber platforms, such as Buxton, Getinge, PBSC, and Spire.
This enables facilities to:
- Upgrade or retrofit existing chamber systems
- Incorporate CURIS Hybrid Hydrogen Peroxide™ technology into new equipment
- Align third-party systems with integrated biodecontamination strategies
Facility Considerations for Chamber Integration |
Implementing a chamber-based biodecontamination system may involve coordination with facility infrastructure, including:
- HVAC operation and airflow management
- Room pressurization and containment
- Door sealing and interlock systems
- Environmental conditions such as temperature and humidity
CURIS works with facility and engineering teams to define requirements and support successful system implementation.
System Scope and Integration Responsibilities 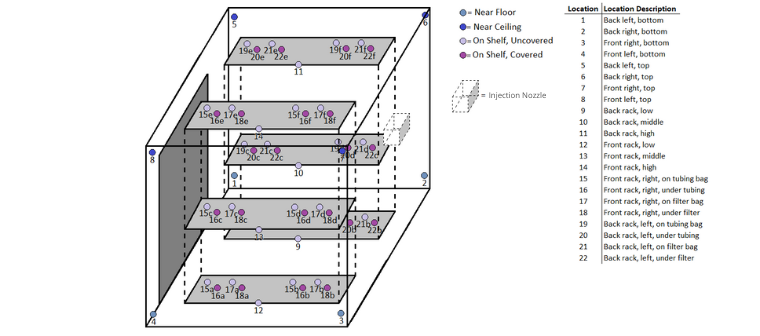
CURIS supports a range of integration approaches depending on facility needs.
Typical CURIS scope includes:
- Biodecontamination system and controls
- Injection and vapor delivery components
- HMI interfaces and reporting systems
Facility or engineering scope may include:
- HVAC and airflow control systems
- Room construction and sealing
- Building automation system integration
CURIS collaborates closely with project teams to ensure alignment across all system components.
Designed for Retrofit and New Construction |
CURIS chamber systems can be implemented in both new and existing facilities:
- Existing material airlocks and transfer spaces
- Retrofit cleanroom environments
- New facility construction projects
In many cases, systems can be deployed without requiring full stainless steel chamber infrastructure or major redesign, depending on application needs.
Designed for Controlled Environments |
CURIS chamber systems support a wide range of controlled environments, including:
Pharmaceutical & Biopharmaceutical Facilities
- Material airlocks for aseptic processing
- Fill-finish and production environments
- Isolator material transfer
 Biosafety & Containment Laboratories
Biosafety & Containment Laboratories
- BSL-2, BSL-3, and BSL-4 environments
- Equipment and material transfer workflows
Vivarium Facilities
- Cage and equipment transfer
- Animal room material movement
Biotechnology & Research Laboratories
- Controlled research environments
- Sensitive equipment and material handling
Explore Integrated Biodecontamination Systems
Chamber systems are one implementation of a broader approach to contamination control.
See how the CURIS® Integration Architecture™ supports integrated biodecontamination across rooms, chambers, and entire facilities.
👉 Explore Integrated Biodecontamination Systems
Request a Chamber Integration Assessment
Discuss your material transfer, throughput, and contamination control requirements with a CURIS specialist.
Identify the right level of chamber integration for your facility—from controlled operation to fully automated workflows.
Trusted by biosafety and pharma leaders around the globe.
All chambers leverage CURIS’ EPA-registered 7% hybrid hydrogen peroxide™ decontamination technology.
Smart, Seamless Integration
Built-In CURIS Technology, Smarter Operation
Rapid Cycles and Aeration
Touchscreen Interface
Seamless Injection
Modular & Mobile Options
The CURIS app allows for remote operation and in-depth data management. Automated systems self-calculate treatment areas, sensors help regulate operation, and users can wirelessly sync up to 20 devices to produce valuable reports of each treatment performed. LEARN MORE